Familial Shar-Pei Fever 3.0
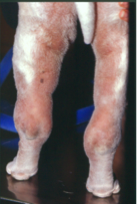
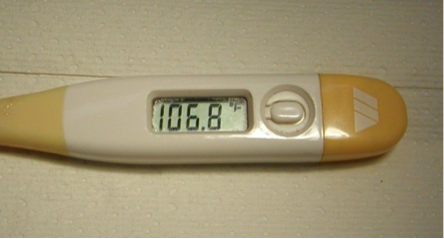
Familial Shar-Pei Fever (FSF) is a hereditary inflammatory disorder seen in Shar-Pei. It appears to be inherited as an autosomal recessive condition. Clinical signs: Episodic fever is the most important and consistent clinical sign of this disorder. The temperature commonly is in the 105-107°F range. The fever is generally self-limiting lasting 12-36 hours. Another common clinical sign often accompanying the fever is swelling of a joint, usually the hock (tibiotarsal) joint and is known as Swollen Hock Syndrome (SHS). This painful, hot swelling can also involve the carpus (wrist) and the lips. Dogs with FSF are sick — they are reluctant to move and when they do walk they have a characteristic “walking on eggs” gait. They often are painful in the abdomen and have a characteristic “roached” back. There can be gastrointestinal signs such as mild vomiting and diarrhea as well.
Pathogenesis: What we do know about this disease is as follows:
- The major function of the immune system is to recognize “SELF” and “NONSELF” and then react appropriately to eliminate the “NONSELF” antigens. There are two arms of the immune system – INNATE IMMUNITY and ADAPTIVE (HUMORAL) IMMUNITY. The innate immune system is set up to react quickly by generating an inflammatory response. The adaptive immune system is slower in onset and has the advantage that it “remembers” previous encounters. Hence it “learns” and reacts quicker and stronger in future episodes. Both these legs work together to lessen the impact of bacteria, foreign material, viruses, fungi, tumor cells, etc. on the body.
- Inflammation is a normal response in the body and is finely controlled by a “checks and balances” system which regulates the duration and intensity of the response. There are systems in place which turn “ON” the system and turn “OFF” the system to maintain balance.
- A genetic defect in the innate immune system can lead to uncontrolled inflammation resulting in AUTOINFLAMMAORY disease – a syndrome of continuous (constant “ON”) inflammation which is not healthy. A genetic defect in the adaptive immune system leads to an over-reactive immune response resulting in AUTOIMMUNE disease (the body reacting to itself).
- FSF is classified as an AUTOINFLAMMATORY DISORDER due to a genetic mutation in innate immunity. There are a number of autoinflammatory diseases besides FSF such as Familial Mediterranean Fever, Tumor necrosis factor Receptor – Associated Periodic Syndrome (TRAPS), Cryopyrin – Associated Periodic Syndrome (CAPS) and others. These are lumped together into Hereditary Recurrent Fever Syndromes (HRFS). These are all diseases which are due to mutations in the genes which regulate the production of inflammatory cytokines (chemical messengers) such as those in the interleukin family. FSF dogs are in a constant state of inflammation with high levels of inflammatory proteins such as IL-6, IL-1β, acute phase reactant proteins, and serum amyloid A (SAA) protein, among others. The development of systemic reactive amyloidosis is a potential complication of all the autoinflammatory diseases.
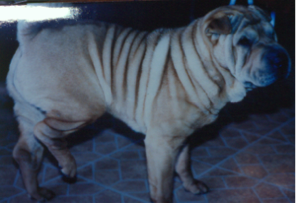
- FSF is more complicated in Shar-Pei due to a genetic mutation which results in HYALURONOSIS – grossly seen as the excessive wrinkles which define the breed. The genetic defect results in over-production of hyaluronan (mucin). Low-molecular weight break down products of hyaluronan metabolism are pro-inflammatory and exacerbate the pro-inflammatory process involved in FSF. Hyaluronan is a normal substance in the body making up the intercellular matrix or scaffolding between cells. Based on its cellular and water content it can present as a free flowing gelatinous material to something as firm as cartilage.
- The tendency to develop amyloidosis may also represent an additional defect that is probably present throughout the canine genome in all breeds of dogs. Amyloidosis develops in the presence of chronic inflammatory diseases such as heartworm, tuberculosis, systemic fungal diseases, chronic bacterial infections and cancer. FSF and hyaluronosis are built-in triggers for the development of amyloidosis and account for the increased susceptibility of the breed to this complication.
- Pedigree studies indicate that FSF is an inherited disorder in the Shar-Pei probably as an autosomal recessive trait. Survey results have indicated that as many as 23% of Shar-Pei experience fevers of unknown origin. Shar-Pei with FSF have increased levels of the cytokine Interleukin-6 (IL-6). IL-6 is involved with the fever response and is an integral part of triggering the production of Acute Phase Reactant Proteins by the liver. IL-6 is also involved in the Systemic Inflammatory Response Syndrome (SIRS). Dysregulation of IL-6 is the cause of much of the disease in Shar-Pei with FSF. IL-6 also plays a major role in the body’s stress response and serves to “prime” the immune system.
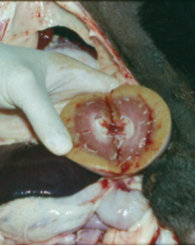
- Shar-Pei with FSF are at risk from early death from systemic amyloidosis. About 5% of the FSF dogs will develop renal failure including renal amyloidosis — a smaller percentage will develop hepatic amyloidosis. This is usually seen in Shar-Pei between the ages of 2-5 years of age. They also seem more susceptible to immune-mediated kidney disease such as membranous glomerulonephritis, protein-losing glomerulopathies, DIC, thromboembolic phenomena such as mesenteric, splenic and pulmonary embolism and Streptococcal Toxic Shock Syndrome (STSS).
- arly on FSF in Shar-Pei was hypothesized to be an animal model for Familial Mediterranean Fever in humans. Recent work indicates this is not true, although FSF is very similar to FMF in man.
Laboratory Findings: Unfortunately there are no blood tests, etc. which are specific for FSF. During a fever episode there will often be an increased white blood cell count, an increase in liver enzyme levels and other non-specific findings. Work done by Dr. Gary Johnson at the University of Missouri College of Veterinary Medicine to develop a DNA blood test to screen for the disease was unsuccessful. The research effort will still continue.
Treatment: It is very important to monitor the temperature in this condition. Initially, fever can be treated using aspirin. Usually a regular strength adult aspirin (325mg) is given every 6- 12 hours for the first 24 hours and then twice a day for 3-5 days thereafter. It is important to continue treatment for several days flowing resolution of the episode to prevent rebound fever. In rare cases where aspirin doesn’t work or for extremely high fevers, dipyrone is given. Other NSAIDs may also be used but response may be a little unpredictable. Some patients will require supportive care with intravenous fluid therapy and in extreme cases emergency treatment similar to heat stroke treatment. Antibiotics are not normally indicated in this condition.
Colchicine: Colchicine is a drug that has been in use in people with FMF to prevent amyloidosis. It is currently being recommended in Shar-Pei with FSF for the same purpose. No studies have been completed to determine if it is useful for this purpose in the Shar-Pei or not. The clinical impression is that it does help. Those dogs on colchicine seem to have fewer FSF episodes and less severe signs while on the drug. Side-effects appear to be minimal at this time. I currently recommend stopping colchicine therapy during an FSF episode while aspirin or other NSAIDs are being used as there is increased risk of gastritis.
Prevention: Shar-Pei with FSF only show symptoms sporadically. It would appear that there are “triggers” involved in initiation of the FSF episodes. One of the major triggers appears to be stress. This may be a dog training class, a dog show, another illness, a dog in heat, excessive exercise, etc. If the owner can recognize these triggers and take steps to avoid them the number of FSF episodes can often be reduced. Diet does not appear to be helpful in prevention of FSF or kidney disease. Surely diet has a role in the management of kidney disease once clinical signs are apparent. Low dose aspirin therapy may be useful in decreasing the incidence of FSF and its severity as well. Aspirin may also be useful as an adjunct therapy in the prevention of thromboembolism.
Monitoring: Monitoring for the complications which often accompany FSF is one of the major goals for the owner of an FSF dog. The primary and most consistent sequela to FSF is kidney failure either due to immune-mediated kidney disease or renal amyloidosis. I currently recommend monitoring a urinalysis every 3 months. The sample should be collected first thing in the morning after the water has been taken up overnight. I primarily look at the urine specific gravity which is a measure of the concentration of the urine and the protein levels in the urine. When the kidneys begin to fail the initial indication is a loss in the ability to produce a concentrated urine. This occurs before there are blood changes related to kidney failure. Increased water consumption, increased urination are the clinical signs associated with a loss of concentrating ability, but these signs are often not recognized. I also think it is wise to do a blood panel every 6-12 months and certainly do one if the urinalysis is abnormal. Weighing your dog periodically is very important. We often don’t recognize a significant weight loss because it is very subtle over a longer period of time. Water consumption and appetite are other important indicators to watch. Amyloidosis is usually medullary in Shar-Pei but can manifest as a protein-losing nephropathy and UPC ratios and treatment with ACE -inhibitors may be necessary.
Complications of FSF: We have already discussed the kidney complications in this condition. Other complications which have been documented include thromboembolism (mesenteric, splenic, pulmonary), DIC (disseminated intravascular coagulation), SIRS (systemic inflammatory response syndrome), MODS (multiple organ dysfunction syndrome), STSS (streptococcal toxic shock syndrome), hypertension associated with renal failure. Many of the deaths following an acute FSF episode are due to these complications. No FSF episode should be treated lightly! Diagnosis: There is no specific diagnostic test for FSF at this time. Diagnosis is based on the clinical sign of episodic fever in a Shar-Pei. I think every Shar-Pei that dies should be autopsied to determine the cause of death, but this is even more critical in cases involving FSF. Renal amyloidosis can only be diagnosed based on kidney biopsy and staining with Congo Red stain. This stain is specific for the presence of amyloid. Amyloid has been found in other tissues in Shar-Pei as well so special staining should be requested on all tissues submitted for histopathology. Many dogs with FSF will not have amyloid in the tissues at the time the tissues were harvested — this means the absence of amyloid in a biopsy specimen does not mean that dog will not or would not have gone on to develop amyloidosis at a later time. To further confuse the issue, not all Shar-Pei with amyloidosis have shown signs of FSF.
Future: Way back in 1983 we first started recognizing episodic fevers in Shar-Pei often associated with a swollen, painful hock joint. Later on it was observed that an early-onset kidney failure occurred in many of these dogs as well. I remember many individuals, including myself, remarking that this has to be associated with the wrinkles in some way and now we know it does.
An important article was recently published in March, 2011, entitled “A Novel Unstable Duplication Upstream of HAS2 Predisposes to a Breed-Defining Skin Phenotype and a Periodic Fever Syndrome in Chinese Shar-Pei Dogs”, Mia Olsson et al. (http://www.plosgenetics.org/article/info:doi/10.1371/journal.pgen.1001332) It is the product of a consortium of researchers from several countries, working in several laboratories and our own Dr. Linda Tintle.
The major points of the article are as follows:
- Chromosome 13
- Using a genome-wide analysis of Shar-Pei and other breeds a breed-specific signal occurred on chromosome 13.
- That same region had the strongest association for susceptibility to FSF.
- Two partially overlapping duplications 14.3 kb and 16.1 kb in size were located which are unique to Shar-Pei and upstream from the HAS2 gene.
- HAS2
- This gene encodes the rate-limiting enzyme synthesizing hyaluronan (HA).
- HA is up-regulated in the folded skin of the Shar-Pei. This is reflected in the higher levels of serum hyaluronan seen in the majority of the breed.
- 16.1 kb duplication
- A high copy number of this duplication results in ↑ expression of HAS2 and ↑ incidence of FSF and/or amyloidosis. It appears this region has a regulatory function and the mutation results in over expression of HAS2.
- Hyper HA syndrome
- ↑copy number variation (CNV) in this mutation predisposes these dogs to FSF.
- Identification of HA as a major risk factor for FSF (and human periodic fevers?).
- Cutaneous mucinosis (Hyper HA syndrome) and FSF are connected and have the same genetic origin – a regulatory gene mutation located close to HAS2.
- ↑HA responsible for the skin condition (excessive folds) predisposes to sterile fever and inflammation.
- Copy number
- A high copy number is not just a genetic marker for FSF but is causally related to the development of disease.
- A higher copy number enhanced the appearance of the phenotype.
- Lowest copy number in Shar-Pei is 5 and the highest is 15 (so far).
- < 4 copies – OK
- 5-10 copies – predominantly FSF
- >10 copies – see FSF and amyloidosis
- 2 alleles involved – total copy number of both alleles
- The mutation occurs as a single copy in other dog breeds (it is a derived mutation in Shar-Pei).
- It is being called “the meatmouth Shar-Pei duplication” because the 16.1 kb duplication appears only in meatmouth Shar-Pei – a type which has elevated levels of HA compared to both traditional Shar-Pei and other breeds.
- Alterations in balance between native high molecular weight HA vs. fragmented low molecular weight HA (including oligomers) may result in activation of the innate immune system.
- Two pathways lead to increased formation of IL-1β, a pro-inflammatory cytokine.
- One pathway involves Toll-like receptors TLR2 and TLR4.
- The other pathway involves the NLRP3 inflammasome – a NLRP3 scaffold, ASC adaptor protein and caspase-1 enzyme. This inflammasome is present in the cytoplasm of monocytes, macrophages and mast cells.
- It is likely a blood test may be available in the near future to screen Shar-Pei for their CNV number and the meatmouth mutation. Breeders can avoid using those individuals with high CNV numbers in their breeding programs. This test may also be useful in the general Shar-Pei population to pick out those individuals who may be at risk for FSF +/- amyloidosis. A possible blood test is being validated at this time.
- A more general conclusion from this paper is the association of degree of wrinkling in American Shar-Pei with the incidence of FSF and amyloidosis. A general guideline would be to only use those individuals with mild to moderate wrinkling in breeding programs and breed away from Hyper HA dogs. It would appear these guidelines are not necessary in the traditional Chinese Shar-Pei.
- Lastly, the association of over-expression of HAS2 and autoinflammation most certainly has implications in the periodic fever syndromes in humans many of which are unexplained at this time. (Jeff Vidt, DVM 3/20/11)
Recommendations: All Shar-Pei with FSF should be on colchicine and be regularly monitored via urine samples and blood work for development of complications. Dogs with FSF should not be used in breeding programs and should be neutered. Dogs with a family history of FSF should be on colchicine and monitored. Dogs with FSF should be maintained as stress-free as possible.